Physical/chemical changes & properties color by number: an innovative approach to unraveling the complexities of matter, its transformations, and the vibrant tapestry of its properties. Embark on a journey where knowledge takes on a kaleidoscope of colors, making learning an immersive and unforgettable experience.
This comprehensive guide delves into the fundamental differences between physical and chemical changes, empowering you with the ability to decipher the subtle nuances that distinguish one from the other. Discover the diverse properties of matter and how they serve as unique identifiers, unraveling the intricate relationship between these properties and the transformations matter undergoes.
Physical and Chemical Changes: Physical/chemical Changes & Properties Color By Number
Physical changes involve changes in the form or appearance of a substance without changing its chemical composition. Chemical changes, on the other hand, involve the rearrangement of atoms to form new substances with different chemical compositions.
Examples of Physical and Chemical Changes
- Melting ice is a physical change.
- Burning wood is a chemical change.
- Dissolving salt in water is a physical change.
- Rusting iron is a chemical change.
Identifying Physical and Chemical Changes
Physical changes can usually be reversed, while chemical changes are usually irreversible. Physical changes do not produce new substances, while chemical changes do. Physical changes involve changes in physical properties, while chemical changes involve changes in chemical properties.
Properties of Matter

Matter has various properties that can be used to identify and classify substances. These properties include:
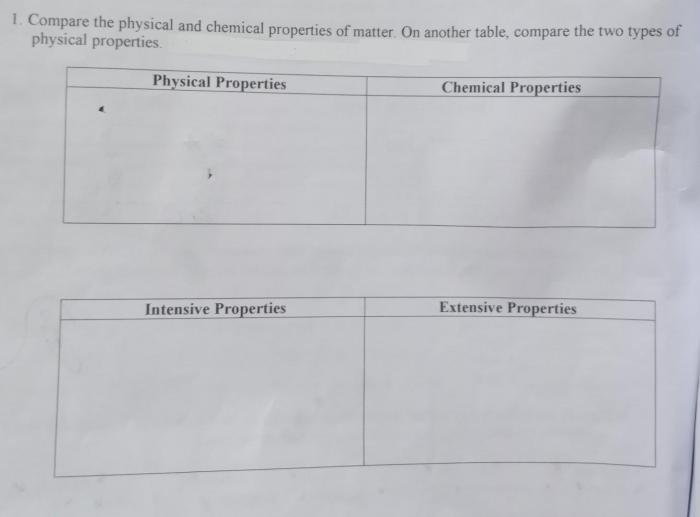
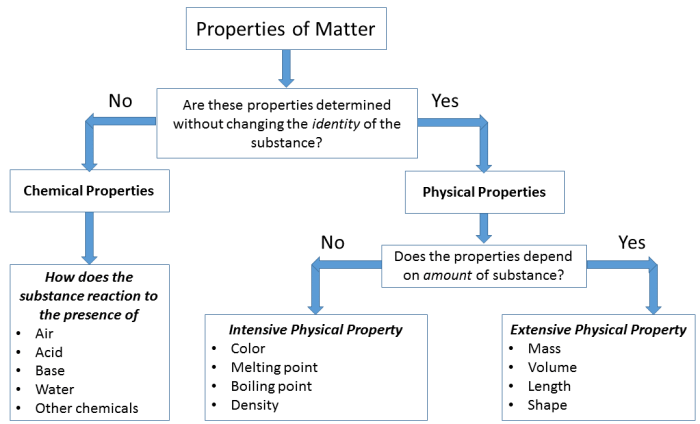
Physical Properties
- Density
- Melting point
- Boiling point
- Solubility
- Color
- Odor
Chemical Properties
- Reactivity
- Flammability
- Acidity/Basicity
- Oxidizing/Reducing ability
The properties of matter are related to its physical and chemical changes. For example, a substance with a low melting point will melt more easily than a substance with a high melting point.
Color by Number
Color by number is a technique that uses colors to represent different physical and chemical properties of matter. This technique can be used to teach about the concepts of physical and chemical changes and the properties of matter.
Benefits of Using Color by Number
- Makes learning more engaging and fun.
- Helps students visualize and understand abstract concepts.
- Reinforces learning through repetition.
Examples of Color by Number Activities, Physical/chemical changes & properties color by number
- Students can color a chart to represent the different physical properties of different substances.
- Students can color a map to represent the different chemical properties of different regions.
- Students can color a diagram to represent the different steps of a chemical reaction.
Activities

Activity 1: Physical and Chemical Changes
Design an activity that uses color by number to teach about physical and chemical changes. For example, students can color a chart to represent the different physical and chemical changes that occur when water is heated.
Activity 2: Properties of Matter
Create a color by number worksheet that teaches about the properties of matter. For example, students can color a chart to represent the different physical properties of different substances.
Activity 3: Color by Number Game
Organize a color by number game that reinforces the concepts of physical and chemical changes and properties of matter. For example, students can play a game where they have to match the color of a substance to its physical or chemical property.
Assessment
Assessment 1: Student Understanding
Develop an assessment that measures students’ understanding of physical and chemical changes and properties of matter. For example, students can take a quiz that tests their knowledge of the different types of physical and chemical changes and the properties of matter.
Assessment 2: Color by Number Activities
Create a rubric that can be used to assess students’ work on color by number activities. For example, the rubric can assess students’ accuracy, creativity, and understanding of the concepts being taught.
Assessment 3: Quiz
Design a quiz that tests students’ knowledge of the concepts covered in this lesson. For example, the quiz can test students’ understanding of the different types of physical and chemical changes, the properties of matter, and how to use color by number to teach about these concepts.
User Queries
What are the key differences between physical and chemical changes?
Physical changes alter the form or appearance of a substance without changing its chemical composition, while chemical changes involve the rearrangement of atoms to form new substances with different properties.
How can color by number be used to teach about the properties of matter?
Color by number assigns different colors to different properties, allowing students to visually identify and compare the properties of various substances.
What are the benefits of using color by number in science education?
Color by number enhances engagement, improves comprehension, and facilitates retention of scientific concepts by making learning more interactive and visually appealing.